Jedtco Medical
SOBIstat-F
SOBIstat-F® Clinical Study Overview
A randomized clinical trial evaluated the SOBIstat-F® device in children experiencing severe acute asthma in the emergency department. The study compared bronchodilator delivery using SOBIstat-F® versus the standard oxygen-assisted method.
Results showed a meaningful improvement with SOBIstat-F®. Children treated with the device had significantly lower hospitalization rates (9.3% vs. 26.8%) and showed faster clinical improvement, including better oxygen levels and pulmonary scores within the first 60–90 minutes. Side effects were similar between both groups, confirming safety.
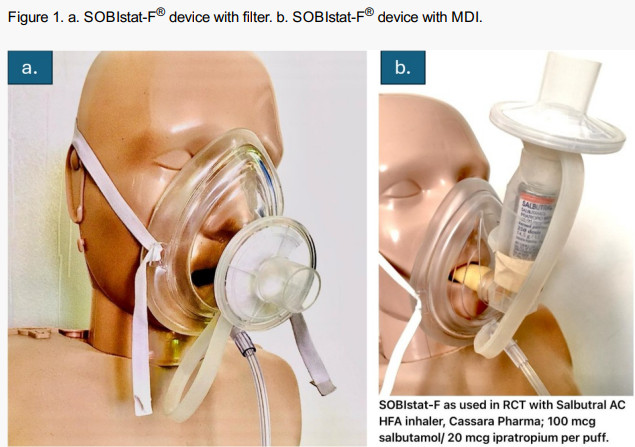
Beyond clinical outcomes, the SOBIstat-F® presentation highlights key practical advantages. The device is designed to deliver both oxygen and medication at the same time without interference, requires minimal setup or training, and can deliver a full therapeutic dose in as little as 2–3 minutes – compared to 10–15 minutes with traditional nebulizers (about 500% faster). It also maintains a closed, filtered system and uses a single mask throughout treatment, improving efficiency in emergency settings .
Overall, SOBIstat-F® offers a faster, easier, and more effective option for treating severe asthma attacks, with strong potential to improve emergency care outcomes.